¶ Rapamycin for Longevity and Anti-Aging
The "Gold Standard" of pharmacological lifespan extension.
¶ Overview

Rapamycin (sirolimus) is a macrolide compound originally discovered in the soil of Easter Island (Rapa Nui) in 1972. Initially developed as an antifungal agent, it was later FDA-approved as an immunosuppressant for organ transplant recipients to prevent rejection. Over the last two decades, it has emerged as the premier candidate for pharmacological lifespan extension, demonstrating robust and reproducible effects across diverse species.
It is currently the only drug consistently proven to extend lifespan in both male and female mice across multiple independent sites in the National Institute on Aging's Interventions Testing Program (ITP). Its mechanism centers on the inhibition of the mechanistic Target of Rapamycin (mTOR), a conserved serine/threonine kinase that functions as a master regulator of cell growth, metabolism, and aging.
¶ Mechanism of Action
The anti-aging effects of rapamycin are mediated through its precise modulation of the mTOR signaling network. This pathway integrates nutrient availability (amino acids, glucose), growth factors, and cellular stress to dictate the cellular switch between anabolism (growth) and catabolism (repair/autophagy).
¶ The mTOR Complexes: mTORC1 vs. mTORC2
mTOR operates in two distinct protein complexes, which differ in function and sensitivity to rapamycin:
- mTORC1 (Rapamycin-Sensitive): This complex acts as the "growth switch." It is the primary regulator of protein synthesis, ribosome biogenesis, and cell proliferation. It is acutely sensitive to rapamycin inhibition.
- mTORC2 (Rapamycin-Insensitive): This complex regulates cytoskeletal organization and insulin signaling (via AKT phosphorylation). It is generally insensitive to acute rapamycin treatment, though chronic exposure can inhibit it indirectly.
¶ Molecular Specificity
Rapamycin acts as an allosteric inhibitor. It does not bind directly to the kinase active site; instead, it binds to the intracellular protein FKBP12. This Rapamycin-FKBP12 complex then binds to the FRB domain of mTORC1, destabilizing it.
- Effect: Inhibition of mTORC1 suppresses the phosphorylation of downstream effectors S6K1 (which drives protein synthesis) and 4E-BP1 (which regulates translation initiation)[1].
- Result: The cell shifts from a "growth" state to a "maintenance" state.
¶ Autophagy Induction
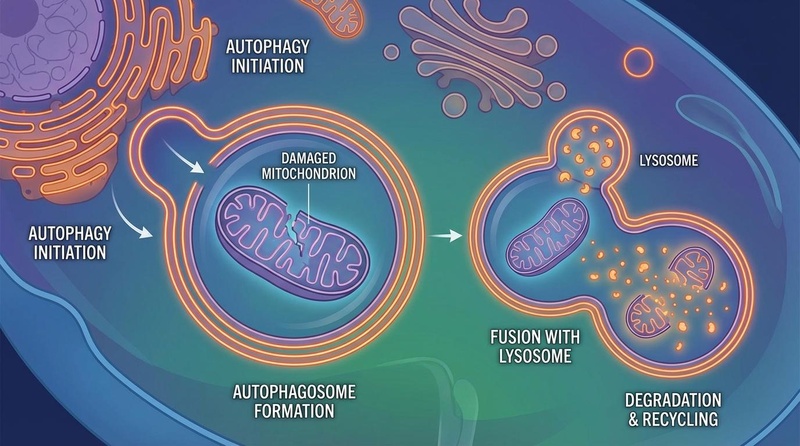
A critical downstream effect of mTORC1 inhibition is the induction of autophagy (cellular recycling). Under nutrient-rich conditions, mTORC1 inhibits the ULK1 complex, blocking autophagy. Rapamycin releases this brake, allowing cells to clear damaged organelles (mitophagy) and misfolded proteins, thereby preserving proteostasis and preventing the accumulation of cellular debris associated with aging[2].
¶ The "Geroconversion" Theory
Dr. Mikhail Blagosklonny's "Hyperfunction Theory of Aging" proposes that aging is a continuation of developmental growth programs. When cells arrest (stop dividing) but mTOR remains active, they do not become quiescent; they become large, hyperactive, and senescent.
- Geroconversion: The transition from cell cycle arrest to senescence.
- Rapamycin as a Gerostatic: By inhibiting mTOR, rapamycin decouples growth from arrest, forcing cells into a reversible quiescent state rather than an irreversible senescent state. This suppresses the Senescence-Associated Secretory Phenotype (SASP), a cocktail of inflammatory cytokines that drives systemic aging[3][4].
¶ Preclinical Evidence (Animal Models)
Rapamycin has the strongest preclinical evidence base of any putative longevity drug, with replicable results across yeast, worms, flies, and mice.
¶ The Interventions Testing Program (ITP)
The National Institute on Aging's ITP is considered the gold standard for longevity testing due to its use of genetically heterogeneous mice (UM-HET3) and parallel testing at three independent sites. Rapamycin has been successful in multiple ITP cohorts:
- 2009 Cohort (Late-Life Start): This landmark study showed that rapamycin extended median lifespan by 23–26% and maximal lifespan by 9–14%, even when treatment started at 600 days (roughly equivalent to a 60-year-old human)[5].
- 2014 Cohort (Dose-Response): Demonstrated that lifespan extension is dose-dependent. At the highest tested dose (42 ppm in food), median lifespan increased by 23% in males and 26% in females. While females showed stronger responses at equivalent dietary concentrations, they also achieved higher blood levels; when adjusted for blood concentration, efficacy appears similar between sexes[6].
- 2020 Cohort (Combinations):
¶ Transient Treatment Effects
A pivotal study by Bitto et al. (2016) challenged the need for chronic daily dosing. Mice treated with high-dose rapamycin for only 3 months in middle age showed a persistent life expectancy increase of up to 60% (relative to remaining life). This suggests that rapamycin induces a stable "remodeling" of the organism—potentially resetting the microbiome or proteome—rather than merely slowing aging while the drug is present[8].
¶ Organ-Specific Rejuvenation
- Heart: Short-term rapamycin treatment (8–10 weeks) can reverse pre-existing age-related cardiac hypertrophy and diastolic dysfunction in old mice, effectively rejuvenating the heart[9].
- Ovaries: Transient treatment in middle-aged female mice delayed ovarian aging, preserving the follicle pool and extending reproductive lifespan well into what would be "post-menopause" for controls[10].
- Brain: In Alzheimer’s models (e.g., hAPP-J20), rapamycin reduces amyloid-beta levels, restores cerebral blood flow, and rescues cognitive deficits[11].
¶ Human Clinical Data
While no human trial has yet measured "lifespan" as a primary endpoint, data from surrogate markers and specific disease states is accumulating.
¶ The Mannick Studies (Immune Function)
Dr. Joan Mannick led Phase 2 trials investigating mTOR inhibitors (everolimus/RAD001 and RTB101) in elderly adults to target immunosenescence.
- Findings: Low-dose mTOR inhibition enhanced the response to influenza vaccination by ~20% and reduced the incidence of total infections compared to placebo[12].
- Significance: These studies overturned the dogma that mTOR inhibitors are purely immunosuppressive. At low doses, they appear to be immunomodulatory, restoring youthful immune function rather than suppressing it.
¶ The PEARL Trial
The Participatory Evaluation of Aging with Rapamycin for Longevity (PEARL) trial is the first large-scale, double-blind, placebo-controlled RCT of rapamycin for healthspan in healthy adults.
- Design: 150+ participants, testing 5mg and 10mg weekly vs placebo over 48 weeks.
- Bioavailability Finding: A crucial finding was that the compounded rapamycin capsules used had ~3.5x lower bioavailability than commercial generic tablets. Thus, the "10mg" group effectively received ~2.9mg of generic equivalent.
- Results (Preliminary 2024):
- Safety: Confirmed safe; adverse events were comparable to placebo.
- Body Composition: Significant preservation of lean muscle mass in women (10mg group).
- Pain: Significant reduction in self-reported pain scores in women.
- Visceral Fat: No significant reduction observed (primary endpoint missed), possibly due to the lower effective dose[13][14].
¶ The Dog Aging Project (TRIAD)
The Test of Rapamycin in Aging Dogs (TRIAD) is an ongoing large-scale veterinary trial. Dogs are an excellent translational model as they share our environment and develop similar age-related comorbidities. Pilot studies have shown safety and improvements in cardiac function (systolic/diastolic) in dogs treated with rapamycin, mirroring mouse data[15].
¶ Dosing Protocols
Note: The following information is derived from clinical trials and medical literature. It is for educational purposes only and does not constitute medical advice.
The "Longevity Protocol" is fundamentally different from the "Transplant Protocol" used in organ recipients.
¶ Daily vs. Weekly Dosing
| Feature | Transplant Protocol | Longevity (Off-Label) Protocol |
|---|---|---|
| Goal | Immune Suppression (Prevent Rejection) | mTORC1 Inhibition (Autophagy/Repair) |
| Frequency | Daily | Weekly / Intermittent |
| Dose | Adjusted to maintain blood trough levels | Fixed low dose (e.g., 5–8 mg/week) |
| mTOR Effect | Inhibits mTORC1 + Chronic mTORC2 inhibition | Pulsed mTORC1 inhibition; spares mTORC2 |
| Side Effects | High risk (Infection, metabolic issues) | Low risk (Mouth sores, mild lipid changes) |
- Transplant Protocol (Daily): High, daily doses are used to maintain constant blood levels, inhibiting both mTORC1 and (eventually) mTORC2 to suppress T-cell proliferation and prevent organ rejection. This carries high risks of infection and metabolic issues.
- Longevity Protocol (Weekly/Pulsed):
- Goal: To inhibit mTORC1 sufficiently to trigger autophagy and repair, while allowing "drug holidays" (troughs) where the drug clears from the system.
- Rationale: The half-life of rapamycin is ~60 hours. Weekly dosing allows mTORC2 to recover before the next dose, minimizing metabolic side effects (insulin resistance) and preventing immune suppression.
¶ Typical Dosages
In off-label usage and trials like PEARL, the most common dosage is 5 mg to 8 mg taken once per week.
- Bioavailability Matters: Generic tablets (e.g., Sirolimus, Rapamune) have higher bioavailability than compounded capsules. Most clinical data is based on the tablet form.
- Absorption: Rapamycin is poorly absorbed; taking it with a fatty meal can increase absorption by ~35%.
¶ Safety and Side Effects
In the context of weekly low-dose administration, rapamycin typically has a mild safety profile, distinct from its reputation in transplant medicine.
¶ Common Adverse Events
- Mouth Sores (Aphthous Stomatitis): The most frequent side effect (occurring in ~15–20% of users). These appear as small, non-infectious ulcers.
- Management: Often transient. Can be treated with dexamethasone mouth rinse or by skipping a dose.
- Metabolic Dysregulation:
- Lipids: Mild elevations in LDL cholesterol and triglycerides are possible. Usually managed with lifestyle or low-dose statins.
- Glucose: While daily dosing causes insulin resistance, weekly dosing generally spares insulin sensitivity. However, monitoring HbA1c is standard practice.
- Bacterial Infection: While viral defense may be improved, rapamycin can inhibit neutrophil function. Caution is advised during active bacterial infections.
¶ Contraindications
- Active Infections: Rapamycin should be paused during active bacterial infections.
- Surgery: As a potent inhibitor of cell proliferation, rapamycin slows wound healing. Standard protocol is to stop usage 2 weeks before and 2 weeks after any major surgery or dental procedure. See [[pages/preventing-scars#surgical-considerations-for-rapamycin-users|Preventing and Minimizing Scars]] for detailed protocols.
- Pregnancy: Rapamycin is teratogenic and must not be used during pregnancy or breastfeeding.
¶ GRADE Assessment
| Outcome | Grade | Evidence Quality |
|---|---|---|
| Lifespan Extension (Mice) | High | Multiple large-scale ITP studies (gold standard), replicated across 3 sites and multiple cohorts. |
| Reversal of Cardiac Aging (Mice/Dogs) | High | Consistent data showing reversal of hypertrophy and improved fractional shortening. |
| Immune Enhancement (Humans) | Moderate | Phase 2b RCTs (Mannick et al.) showing improved vaccine response and reduced infections. |
| Muscle Preservation (Humans) | Moderate | PEARL trial showed significant lean mass preservation in women. |
| Lifespan Extension (Humans) | Very Low | No direct lifespan data exists. Inferred from biology and surrogate markers. |
¶ References
Saxton RA, Sabatini DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell. 2017;168(6):960-976. ↩︎
Kaeberlein M. Rapamycin and cancer: In search of the magic bullet. Science. 2010. ↩︎
Blagosklonny MV. Rapamycin for longevity: opinion article. Aging (Albany NY). 2019;11(19):8048-8067. ↩︎
Blagosklonny MV. Cell senescence, rapamycin and hyperfunction theory of aging. Cell Cycle. 2022;21(14):1456-1467. ↩︎
Harrison DE, et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature. 2009;460(7253):392-395. ↩︎
Miller RA, et al. Rapamycin-mediated lifespan increase in mice is dose and sex-dependent and metabolically distinct from dietary restriction. Aging Cell. 2014;13(3):468-477. ↩︎
Strong R, et al. Lifespan benefits for the combination of rapamycin plus acarbose... in genetically heterogeneous mice. Aging Cell. 2020. ↩︎
Bitto A, et al. Transient rapamycin treatment can increase lifespan and healthspan in middle-aged mice. eLife. 2016;5:e16351. ↩︎
Flynn JM, et al. Late-life rapamycin treatment reverses age-related heart dysfunction. Aging Cell. 2013;12(5):851-862. ↩︎
Dou X, et al. Short-term rapamycin treatment increases ovarian lifespan in young and middle-aged female mice. Aging Cell. 2017;16(4):825-836. ↩︎
Lin AL, et al. Chronic rapamycin restores brain vascular integrity and function through NO synthase activation and improves memory in symptomatic mice modeling Alzheimer's disease. J Cereb Blood Flow Metab. 2013. ↩︎
Mannick JB, et al. mTOR inhibition improves immune function in the elderly. Science Translational Medicine. 2014;6(268):268ra179. ↩︎
Strong R, et al. Preliminary Results of the PEARL Trial. MedRxiv. 2024. ↩︎
Fairfield B. Results of the PEARL Trial: An Expert Analysis. AgelessRx. 2024. ↩︎
Urfer SR, et al. A randomized controlled trial to establish effects of short-term rapamycin treatment in 24 middle-aged companion dogs. GeroScience. 2017;39(2):117-127. ↩︎